Empirical Formula of Benzene
Using the above formula ε 36600 for the 395 nm peak and 14000 for the 255 nm peak. Therefore the degree of unsaturation of benzene is 4 which gives one.

C6h6 Lewis Structure Benzene Lewis Chemical Formula Home Decor Decals
Calculate the molecular weight of the compound.

. Benzene was first isolated by Michael Faraday in 1825 from the whale oil used in gaslights. Benzene a 352 toluene a 613 ethy lbenz ene a 1065 o-xy lene a 1292 m-xy lene a 1133 p-xy lene a 1104 cumene b 1861 p-ter t-bu ty lt oluene b 2145 -methyl sty rene b 1999 -methyl sty rene b 2082 styrene b 1833 a Separation achieved using a 30-m Stabilwax fused silica capillary colum. Methocarbamol is a white powder sparingly soluble in water and chloroform soluble in alcohol only with heating and propylene glycol and insoluble in benzene and n-hexane.
Separate multiple products using the sign from the drop-down menu. Its molecular weight is 24124. First select Enthalpy Formula option.
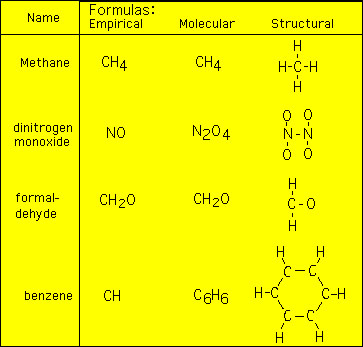
Eilhardt Mitscherlich synthesized benzene in 1834 and showed it to have a molecular formula of C 6 H 6Many other compounds with similar properties to benzene were discovered in the 1800s all. Then we divide by the smallest number to get the ratios of each element. Empirical Formula - Meaning Examples Statistics and FAQs.
A molecular formula provides more information about a molecule than its empirical formula but is more difficult to establish. Draw a structural formula for the substitution product of the reaction shown below. If more than one stereoisomer of product is formed draw both.
F CH3 Na OČCH3 CHCO2H Use the wedgehash bond tools to indicate stereochemistry where it exists. Caffeine Chemical Formula - Meaning Properties and FAQs. The chemical name of methocarbamol is 3-2-methoxyphenoxy -12propanediol 1-carbamate and has the empirical formula C 11 H 15 NO 5.
For the spectrum on the right a solution of 0249 mg of the unsaturated aldehyde in 95 ethanol 142 10-5 M was placed in a 1 cm cuvette for measurement. Calculate empirical formula and molecular formula. With the chart below the empirical formula of the compound can be used to determine solubility by cross referencing the cations top row with anions first column.
The enthalpy calculator calculates. 6131 g x 5105 mol C. 1365 g of an unknown compound decreased the freezing point of 346 g of benzene Kf 512 C m by 129 C.
For water both formulae are H 2 O. B Separation achieved using a 30-m Rtx-35 fuse d. Empirical Formula - Meaning Examples Statistics and FAQs.
Then put values against each parameter in tool and calculate. One can notice that all these have the exact same end to the molecule. The only difference is the complexity of the other attached group.
In aldehydes the carbonyl group has one hydrogen atom attached to it together with either a 2nd hydrogen atom or a hydrogen group which may be an alkyl group or one containing a benzene ring. The structural formula is shown below. After doing so select either Internal energy volume or Change in internal energy volume.
Unlike other chemical formula types which have a limited. The structural formula is shown below. Ebullioscopic constant for benzene.
However the question gives the molar mass as text78 textgmol-1. A solution of 100 g of a nonvolatile non-dissociating compound dissolved in 0200 kg of benzene boils at 812 C. The molecular formula of benzene is C₆H₆.
The molar mass of benzene using the empirical formula is text13 textgmol-1. He also determined that it had an empirical formula of CH. Thus it has six carbon atoms and it needs 8 more hydrogen atoms in order to be classified as saturated.
The structural formula of a chemical compound is a graphic representation of the molecular structure determined by structural chemistry methods showing how the atoms are possibly arranged in the real three-dimensional spaceThe chemical bonding within the molecule is also shown either explicitly or implicitly. Ethanol acetone methyl acetate ethyl acetate diethyl ether. An example of the difference is the empirical formula for glucose which is CH 2 O ratio 121 while its molecular formula is C 6 H 12 O 6 number of atoms 6126.
For less common compounds you can consult a periodic table while using the solubility guidelines listed above. Stoichiometry Calculating Empirical Formulas Assuming 10000 g of para-aminobenzoic acid C. Its molecular weight is 24124.
Benzene reacts with 65 g of bromine and produces 567 g of bromobenzene what is the percent yield of the reaction. To find the empirical formula we first note how many moles of each element we have. Empirical formula of PABA.
253 Cm and Boiling Point of pure benzene. Its molecular weight is 24124. What is the molar mass of the compound.
What Is The Empirical Formula For Benzene C6h6 Quora

Aromatic Compounds And Electrophilic Aromatic Substitution Concise Medical Knowledge Organic Chemistry Benzene Chemistry Lessons

The Empirical Formula Of Benzene And Acetylene Is Are Youtube
Quantitative Chemistry Molecular Formulas
0 Response to "Empirical Formula of Benzene"
Post a Comment